The safety assessment of the Canadian nuclear waste disposal program requires a fundamental understanding of the processes controlling fuel corrosion which could lead to the release of radionuclides to the geosphere from a failed container. The redox conditions controlling fuel (UO2) corrosion inside a failed container will be dictated by the alpha radiation fields within the fuel and their influence will change the composition of the UO2 surface. This influence of radiation is being simulated electrochemically. The composition of the surface can be determined by X-ray photoelectron spectroscopy (XPS) and the extent of surface damage due to dissolution observed by scanning electron microscopy (SEM).
The influence of groundwater composition, in particular carbonate/bicarbonate, has been evaluated. In the figures below, the XPS spectra were deconvoluted to show the fractions of three uranium oxidation states (U(IV), U(V) and U(VI)) present on (a) a freshly polished UO2 surface and after anodic oxidation in a solution without (b) and with (c) HCO3(-)/CO3(2-). Combined with the SEM images and additional electrochemical data, it was shown that the presence of HCO3(-)/CO3(2-) facilitates the dissolution of insulating U(VI) species, exposing the underlying U(IV)/U(V) species which are catalytic and significantly affect the surface redox reaction rates and alter the overall fuel corrosion rate.
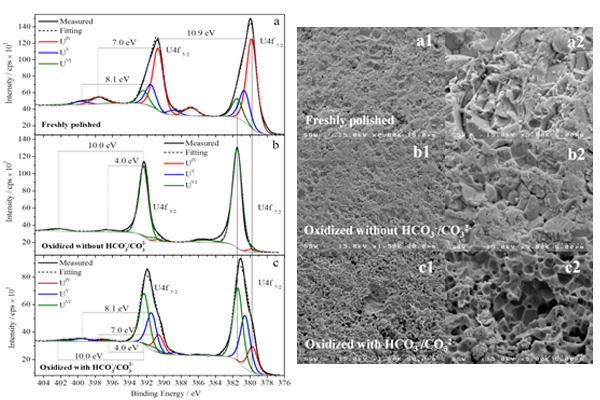
Figures: The XPS spectra (left) showing the deconvoluted U4f5/2 and U4f7/2 regions and the corresponding SEM micrographs (right) recorded on a simulated nuclear fuel surface before and after anodic oxidation at E = 0.30 V (vs. SCE) for 0.5 hour in 0.1 mol/L NaCl + 0.02 mol/L H2O2 at pH 11.0 with and without HCO3(–)/CO3(2–).
By Linda (Michael) Wu, Dept. of Chemistry, Western University

